Saturday, April 4, 2026
← Back to archivePolicy
Policy•Fierce Pharma•Apr 3, 2026

Fierce Pharma Asia—Trump’s 100% drug tariff; Takeda layoffs; Lilly, Insilico's AI deal
background ▾
- •Trump's 100% drug tariff creates significant regulatory uncertainty for pharma supply chains, though orphan drugs, specialty medicines, and MFN agreements are exempt, and country-specific trade deals supersede the tariff.
- •Eli Lilly commits up to $2.75B to Insilico Medicine for AI-driven drug discovery with exclusive worldwide license to oral therapeutics, signaling major pharma's deepening investment in AI platforms.
- •Monitor implementation details on tariff enforcement, which companies qualify for exemptions, and potential retaliatory trade actions that could destabilize existing manufacturing and sourcing agreements across the industry.
Eli LillyInsilico MedicineTakedaMDMA analogGLP-1
→ Read original articlePolicy•Fierce Pharma•Apr 3, 2026

An update on the pharma industry’s reshoring effort
background ▾
- •Pharma industry has pledged ~$370B in U.S. investments with high-profile projects like UCB's $2B Atlanta biologics plant, but near-term domestic production impact will be limited
- •Reshoring is being driven by both supply chain resilience objectives and 2025 tariff threats, with companies front-loading manufacturing in response to trade policy uncertainty
- •Watch for: actual facility completion timelines, whether announced investments materialize, tariff policy evolution, and whether domestic production capacity actually offsets import dependencies
UCBUnspecified pharma companies (industry-wide trend)
→ Read original articlePolicy•Fierce Biotech•Apr 3, 2026

White House floats 12.5% budget cut for HHS in FY2027, reiterates reorganization plan
background ▾
- •NIH faces a proposed $5 billion cut with elimination of three institutes (National Institute on Minority Health and Health Disparities, Fogarty International Center, National Center for Complementary and Integrative Health), directly threatening fundamental research infrastructure and grant funding that biotech/pharma relies on for early-stage development.
- •The proposed Administration for a Healthy America reorganization would consolidate CDC, HRSA, and SAMHSA functions and claims $5 billion in savings through program elimination—a structural change Congress rejected last year but the administration is pursuing more aggressively this cycle.
- •Monitor Congressional response in spring 2026 budget negotiations; pharma should assess impact on NIH grant funding, clinical trial infrastructure, and regulatory modernization plans, particularly the AI-modernization priority that could reshape FDA operations.
Policy•Fierce Biotech•Apr 2, 2026

Trump slaps 100% duties on imported drugs but leaves plenty of exceptions
background ▾
- •100% tariffs on patented drugs and ingredients begin in 120-180 days, but MFN-agreement companies are fully exempt until January 2029, creating a two-tiered system that favors large pharma
- •Generics, biosimilars, orphan drugs, and products from EU/Japan/Korea/Switzerland are exempt or face reduced rates; new UK trade deal provides full exemption for British drug exports
- •Mid-size biotech companies without MFN agreements face significant competitive disadvantage; White House is now negotiating MFN deals with smaller drugmakers, suggesting policy may evolve based on industry pressure
Biotechnology Innovation OrganizationMidsized Biotech Alliance of America
→ Read original articlePolicy•KFF•Apr 2, 2026
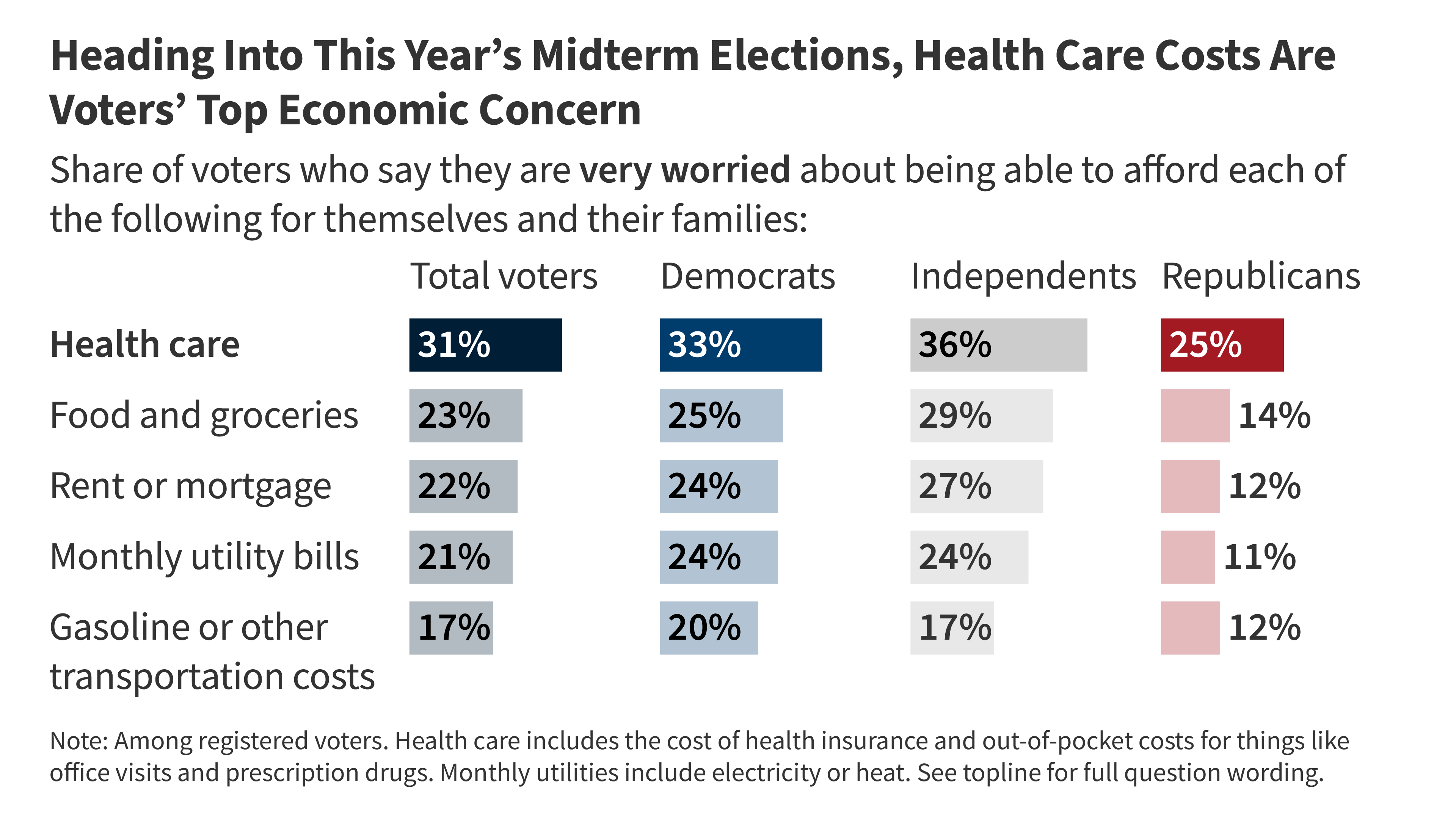
A Preview of the Role Health Care May Play in the 2026 Election
background ▾
- •Health care costs function as a bridge between health care and economic voter concerns, making them a critical factor in election strategy regardless of how the issue is framed
Market Access
Market Access•Fierce Biotech•Apr 3, 2026

ADCs, TCEs among ‘exciting things’ on Boehringer’s dealmaking shopping list
background ▾
- •Boehringer has committed >$3 billion to licensing deals in 2026 alone (Variant Bio, Rectify, Simcere, Sitryx, AimedBio from 2025), signaling aggressive portfolio expansion and significant capital allocation to external innovation.
- •The company explicitly prioritizes preclinical candidates to reduce acquisition costs and maintain strategic flexibility, exploiting a 'seller's market' where late-stage assets command premium valuations.
- •Watch for: announcements of TCE and ADC licensing deals; readouts from obrixtamig phase 3 in small-cell lung cancer; expansion of the Swiss ADC R&D facility; and clinical-stage data from in-vivo CAR-T programs in autoimmune indications.
Boehringer IngelheimVariant BioRectify Pharmaceuticalsobrixtamig
→ Read original articleMarket Access•Fierce Biotech•Apr 3, 2026

Months after $100M series A, Syneron closes $150M round to support peptide platform
background ▾
- •Syneron has now raised >$250M in less than 6 months, demonstrating exceptional investor appetite for macrocyclic peptide platforms and validating the commercial viability of the 'goldilocks' drug class
- •The AstraZeneca partnership ($75M upfront + $3.4B potential milestones) combined with Series B funding suggests Syneron's platform has achieved significant technical/clinical validation that justifies mega-deal economics
- •Watch for: clinical trial initiation timelines in oncology and autoimmune programs; competitive positioning relative to Unnatural Products and other macrocyclic peptide developers; and potential additional pharma partnerships leveraging the platform
Syneron BioAstraZenecaDecheng Capital
→ Read original articleMarket Access•Fierce Pharma•Apr 3, 2026

Biopharma R&D pipeline shrinks for 1st time in 30 years: report
background ▾
- •Global drug development pipeline contracted to 22,940 molecules in early 2026, breaking 30-year growth trend, with new drug entries falling 1.3% in 2025
- •Big Pharma restructuring: Roche now #1 (262 drugs), Pfizer dropped to #3 after cutting 14 programs, AstraZeneca surged to #2, signaling deliberate pipeline rationalization among tier-1 companies
- •Neurological disease rising sharply as strategic priority, growing from 12.7% of new pipeline entries (2023) to 14.4% (2025), while oncology maintains dominance at 38.6% of new programs
RocheAstraZenecaPfizer
→ Read original articleClinical Trials
Clinical Trials•Fierce Biotech•Apr 2, 2026

Buoyed by $100M Series A, Stipple Bio debuts to advance lead oncology asset into clinic
background ▾
- •Stipple's Pointillist platform represents a differentiated approach to identifying tumor-specific epitopes, positioning the company to build a multi-asset oncology pipeline beyond its lead ADC program
- •The $100M Series A (expected to fund through 2029) signals strong investor confidence and provides substantial runway, though clinical validation of STP-100's safety and efficacy advantage remains unproven
Stipple BioRA Capitala16z Bio+HealthSTP-100
→ Read original article