Friday, May 29, 2026
Today at a Glance10 articles · 4 sources
Top Stories
- 1
- 2Hoth Therapeutics’ pivot to AI in space sees stock rocket after rebrandMarket AccessFierce Biotech
- 3Novartis ‘always looking’ for further ways to shuttle drugs to brain despite run of recent dealsMarket AccessFierce Biotech
Key Entities
Companies
Beren TherapeuticsVtesseSucampoMallinckrodtMandosNational Institutes of Health
Drugs & Therapies
ZepboundadrabetadexElaheremirvetuximab soravtansinesemaglutide
Coverage
- Policy5
- Market Access5
- Clinical Trials0
Sources
Fierce Biotech, Fierce Pharma, STAT News, KFF
Policy
Policy•Fierce Biotech•May 28, 2026

FDA extends Beren’s rare disease review after info request, further stretching saga
background ▾
- •FDA classified Beren's data response as a major amendment, triggering a three-month extension of the review clock and signaling potential substantive deficiencies in the original filing.
- •The approval case rests entirely on OS comparison to external controls (84% vs. 42% at 5 years) rather than a randomized trial, which exposes the application to rejection given FDA's recent skepticism of natural history arms in rare disease approvals.
- •Monitor the November 17 decision date and any interim communications from Beren; rejection would likely require a new trial or stronger real-world evidence to overcome FDA's methodological concerns.
Beren TherapeuticsVtesseSucampoadrabetadex
→ Read original articlePolicy•Fierce Pharma•May 29, 2026
Pharma leaders meet with PM Takaichi in push for Japan to retain R&D edge
background ▾
- •Global pharma executives are directly engaging with Japan's top leadership to reverse the country's declining biopharma R&D footprint and competitiveness.
- •Japan's share of global pharmaceutical research and development has been eroding, prompting industry to seek government policy support.
- •Watch for announcements of specific Japanese government policy changes around R&D incentives, tax benefits, regulatory streamlining, or innovation funding in coming months.
Policy•Fierce Pharma•May 29, 2026
With Elahere building steam, AbbVie nets FDA nod for another ImmunoGen cancer asset
background ▾
- •AbbVie secures second FDA approval from ImmunoGen acquisition, strengthening ADC portfolio strategy
- •Elahere momentum combined with this new approval signals successful execution of ImmunoGen deal integration
- •Watch for commercial uptake trajectory of the newly approved asset and pipeline progression of remaining ImmunoGen candidates
AbbVieImmunoGenElaheremirvetuximab soravtansine
→ Read original articlePolicy•STAT News•May 29, 2026

Opinion: I’m a weight-loss doctor. Here’s why I worry about GLP-1 ‘microdoses’
background ▾
- •Microdosing GLP-1s for weight loss lacks any rigorous clinical evidence and is primarily driven by direct-to-consumer compounded products without FDA approval, distinct from legitimate dose adjustments by medical professionals
- •FDA-approved GLP-1 therapies (semaglutide doses: 0.25–2.4 mg weekly or 1.5–25 mg daily; tirzepatide doses: 2.5–15 mg weekly) are the only doses rigorously studied for safety and efficacy; off-label microdosing exposes patients to unknown long-term risks
- •Watch for regulatory action against compounded GLP-1 products, physician-led guidelines on maintenance-phase dosing, and potential CMS/payer responses to off-label use patterns
semaglutideWegovyOzempic
→ Read original articlePolicy•KFF•May 28, 2026
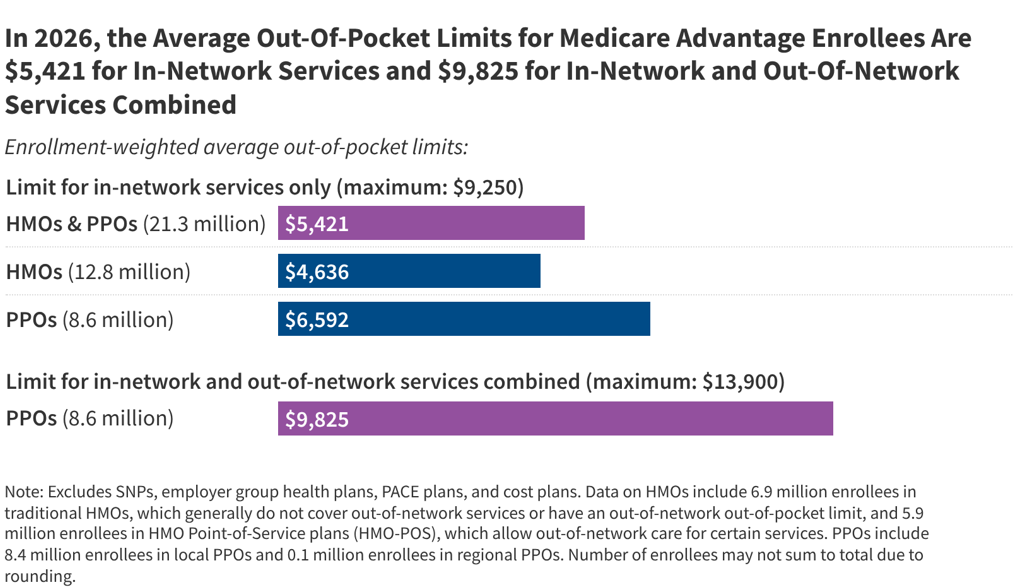
Medicare Advantage Out-of-Pocket Limits: Variation and Trends
background ▾
- •Medicare Advantage enrollees face substantial variation in out-of-pocket limits depending on plan type and state, with average limits significantly below statutory maximums, reflecting how plans allocate federal rebate dollars
Market Access
Market Access•Fierce Biotech•May 28, 2026

Hoth Therapeutics’ pivot to AI in space sees stock rocket after rebrand
background ▾
- •Hoth Therapeutics has completely exited biopharmaceutical development and rebranded as Rocket One to pursue AI chip technology for orbital applications, with stock doubling on announcement.
- •The company's previous phase 2 data for an EGFR inhibitor-induced skin toxicity treatment and obesity-focused pipeline failed to attract investor support, prompting the drastic strategic pivot.
Hoth Therapeutics / Rocket OneSpaceXneurokinin 1 receptor agonist (EGFR inhibitor-induced skin toxicities)parenteral glial cell-derived neurotrophic factor
→ Read original articleMarket Access•Fierce Biotech•May 28, 2026

Novartis ‘always looking’ for further ways to shuttle drugs to brain despite run of recent deals
background ▾
- •Novartis' $12B Avidity acquisition gives it three clinical-stage TFR-based therapies with demonstrated target engagement, including del-zota (DMD) approaching regulatory submission with 25% dystrophin increase data
- •The company is actively translating proven TFR muscle-targeting technology to neurodegenerative indications (Alzheimer's, Parkinson's) via partnerships with SciNeuro ($165M) and Arrowhead ($200M), though brain BBB tuning is acknowledged as more complex than muscle delivery
- •Competitive landscape intensifying: Roche advancing its own brain shuttle bispecific in Phase 3 and partnering with Manifold Bio; clinical readouts from Novartis' muscle programs in 2025 will signal likelihood of successful brain translatability and inform valuation of its broader BBB platform
NovartisSciNeuro PharmaceuticalsRochedelpacibart zotadirsendelpacibart etedesirandelpacibart braxlosiran
→ Read original articleMarket Access•Fierce Pharma•May 29, 2026
AstraZeneca gains 2nd bladder cancer nod in key expansion for Imfinzi
background ▾
- •Imfinzi becomes the first-to-market PD-L1 inhibitor combination regimen approved for high-risk NMIBC, establishing a differentiated position versus Keytruda monotherapy in this patient population
- •This approval adds to AstraZeneca's growing bladder cancer franchise and demonstrates momentum in checkpoint inhibitor combinations across multiple indications
- •Monitor real-world uptake, payer reimbursement decisions, and whether Merck pursues combination strategies to remain competitive in the NMIBC segment
AstraZenecaMerckImfinzidurvalumabKeytruda
→ Read original articleMarket Access•Fierce Pharma•May 29, 2026
CVS restores coverage of Eli Lilly obesity med Zepbound, adds new pill Foundayo
background ▾
- •CVS Caremark's restoration of Zepbound coverage and addition of Foundayo removes a major market access barrier for Eli Lilly's obesity franchise, potentially affecting millions of insured patients
- •The move signals potential softening of PBM coverage restrictions on obesity medications, though formulary status and any associated restrictions (prior authorization, step therapy) remain unclear
- •Competitive dynamics with Novo Nordisk's Ozempic/Wegovy and other GLP-1 agents will likely continue to drive PBM coverage negotiations and formulary positioning
CVS CaremarkEli LillyZepboundFoundayo
→ Read original articleMarket Access•Fierce Pharma•May 29, 2026
PharmaEssentia taps Incyte alum Eric Vogel as it eyes Besremi expansion
background ▾
- •PharmaEssentia is actively pursuing label expansion for Besremi with a dedicated commercialization leadership hire from a major oncology-focused pharma company
- •The appointment of a commercialization head with Incyte background suggests the company is preparing for multi-indication strategy execution
PharmaEssentiaIncyteBesremi
→ Read original articleClinical Trials
No articles selected for this category today.