Thursday, April 2, 2026
← Back to archivePolicy
Policy•Fierce Biotech•Apr 1, 2026

Whoop raises $575M series G, Abbott comes on board amid hiring spree
background ▾
- •FDA reversed July 2024 warning letter in January 2025, exempting Whoop's wearable from medical device classification and allowing it to operate under general wellness framework—a watershed regulatory decision
- •Abbott's participation signals major pharma/medtech validation of Whoop's platform and potential future integration with established healthcare ecosystems
- •Valuation jumped to $10.1B on strong investor conviction, but commercial model depends on sustained subscriber growth in competitive fitness wearable market
WhoopAbbottMayo Clinic
→ Read original articlePolicy•Fierce Pharma•Apr 1, 2026

FDA extends review of Orca Bio’s novel cell therapy for blood cancers
background ▾
- •FDA regulatory scrutiny of cell therapy manufacturing and controls is intensifying across the industry, as evidenced by repeated CMC-related delays and the planned departure of CBER director Vinay Prasad, M.D.
- •Orca-T's clinical efficacy is not in question—the phase 3 data are robust—but the CMC extension underscores the manufacturing complexity of allogeneic cell therapies and the FDA's tightened standards for product specifications and potency assays.
- •Monitor the July 2026 FDA decision and watch for whether the incoming CBER leadership signals any policy shifts on cell therapy review standards; also track uptake and safety signals from Orca Bio's expanded access program.
Orca BioReplimuneOrca-TRP1
→ Read original articlePolicy•KFF•Apr 1, 2026
Policy Tracker: Exceptions to State Abortion Bans and Early Gestational Limits
background ▾
- •20 US states now have abortion bans or early gestational restrictions in effect, with varying exception categories that directly impact access to reproductive healthcare and potential pharmaceutical/procedural interventions
Policy•KFF•Apr 1, 2026
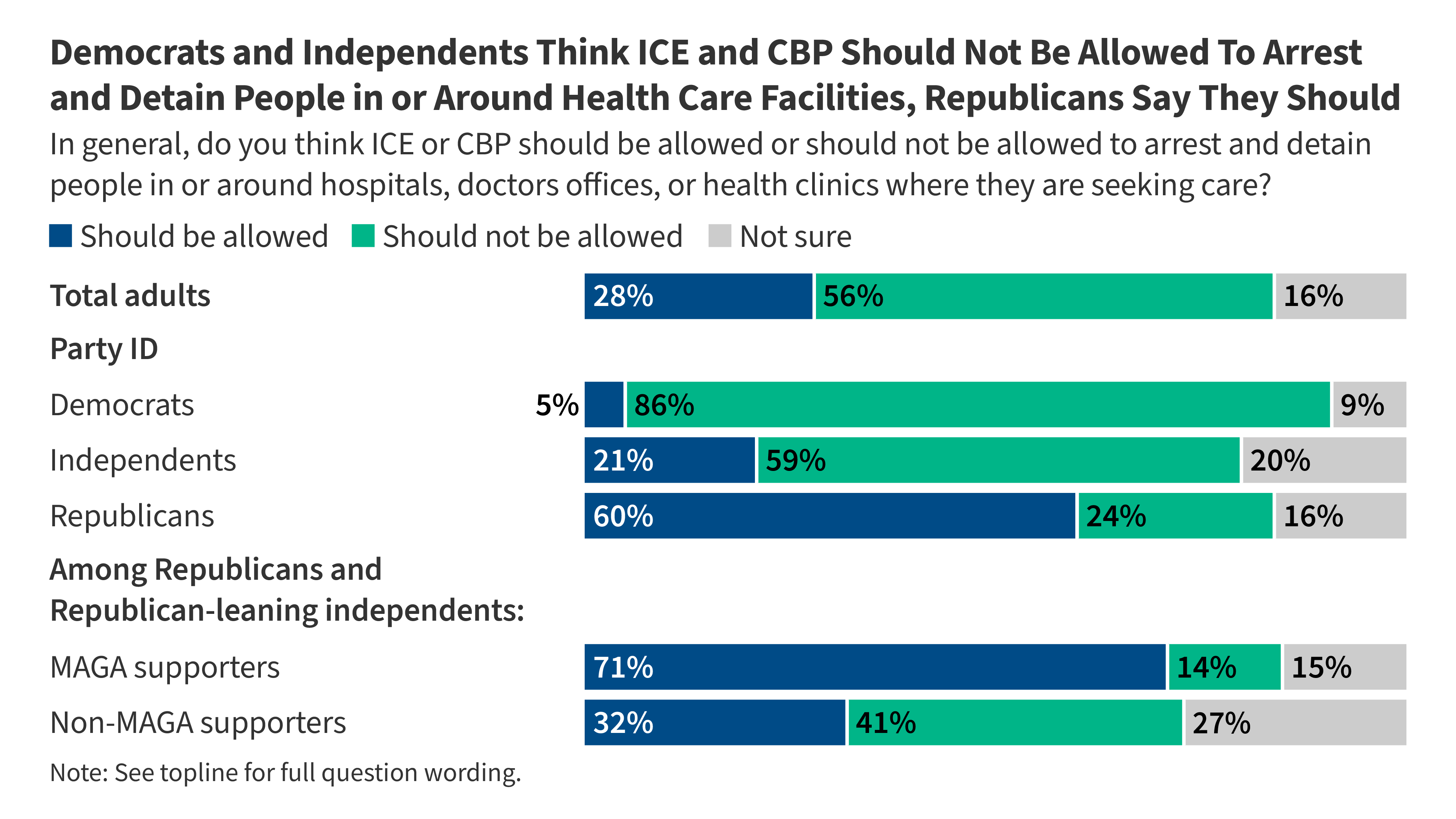
KFF Health Tracking Poll: The Public’s View of Immigration Enforcement Activities in Health Care Settings
background ▾
- •This is a public opinion poll with no direct pharmaceutical industry, drug development, or market access implications
Policy•Fierce Pharma•Apr 1, 2026

‘There isn't as much meat left to cut’: Biopharma layoffs maintain slowdown in Q1
background ▾
- •Q1 2026 saw 33 biopharma layoff announcements—the lowest quarterly count since Q1 2022 and less than half the 63 rounds reported in Q1 2025, indicating a deceleration of industry workforce reductions.
- •Large pharma companies have largely exhausted restructuring opportunities, with experts suggesting the wave of mass layoffs may be subsiding as strategic realignment completes, though clinical failures continue to trigger biotech job cuts.
- •Notable Q1 2026 restructurings included Takeda (634 U.S. jobs), Evotec (800 jobs across site closures), and severe biotech reductions at Theravance (50% headcount reduction) and Gossamer Bio (48% reduction) driven by phase 3 failures.
Novo NordiskBayerTakedaTrintellixseralutinib
→ Read original articleMarket Access
Market Access•Fierce Biotech•Apr 1, 2026

Alzheimer's-focused Korsana to use Cyclerion reverse merger to go public
background ▾
- •Korsana has secured $555 million total funding ($175M + $380M) and is taking a competitive transferrin-receptor-binding approach to Alzheimer's—a mechanism also being pursued by Roche (trontinemab) and AbbVie (via Aliada acquisition)
- •The reverse merger with Cyclerion provides immediate public company status and capital runway to advance KRSA-028 into phase 1, with clinical data readout timeline of end-2027
- •Watch for phase 1 enrollment and early amyloid plaque clearance data in KRSA-028; monitor competitive landscape as transferrin-binding becomes crowded (Roche in phase 3, AbbVie asset in development)
Korsana BiosciencesCyclerion TherapeuticsFairmountKRSA-028trontinemab
→ Read original articleMarket Access•Fierce Pharma•Apr 1, 2026

Lilly answers Novo's GLP-1 pill with highly anticipated FDA nod for Foundayo
background ▾
- •Foundayo's approval ends Novo's monopoly on oral GLP-1s for obesity and introduces meaningful price competition ($149/month vs. Wegovy's matched pricing) in a rapidly expanding market.
- •ATTAIN-1 data demonstrates superior weight loss efficacy (12.4% vs. Wegovy's 16.6%), though lacking head-to-head comparison and unclear real-world applicability across different patient populations.
- •Watch for uptake trajectories, payer coverage decisions, and whether Novo's cardiovascular outcome data advantage (LEADER trial) becomes differentiating in market positioning and reimbursement leverage.
Eli LillyNovo NordiskFoundayo (orforglipron)Wegovy (semaglutide)
→ Read original articleMarket Access•Fierce Pharma•Apr 1, 2026

Novo's Wegovy nets cardio nod from UK cost gatekeeper, adding 1M+ eligible patients
background ▾
- •NICE approval expands Wegovy's addressable market in the UK by 1.2 million patients with established cardiovascular disease, creating a distinct clinical positioning separate from obesity indication
- •SELECT trial data demonstrated cardiovascular benefit occurs independent of significant weight loss, suggesting direct cardioprotective mechanisms—a differentiation point versus weight-loss-dependent competitors
- •NHS implementation timeline of summer 2024 accelerates commercial availability and de-risks reimbursement uncertainty, but Novo still faces intense US market competition from Eli Lilly and emerging generic pressure in India
Novo NordiskEli LillyWegovysemaglutideOzempic
→ Read original articleClinical Trials
Clinical Trials•Fierce Biotech•Apr 1, 2026

Gilead cancels midstage HIV trial as FDA’s clinical hold remains intact
background ▾
- •Gilead terminated the Wonders-2 trial after 13 months of FDA clinical hold; the broader hold on all related studies remains in place, indicating unresolved regulatory concerns
- •CD4+ T-cell and white blood cell toxicity prompted the original hold, though values have normalized—the persistence of the FDA hold suggests safety concerns may extend beyond the observed hematologic effects
- •Monitor the status of Wonders-1 (675 participants still enrolled) and the three phase 1 studies; any FDA guidance on trial resumption or a decision to uphold the clinical hold indefinitely would signal whether GS-1720/GS-4182 combination therapy remains viable
Gilead SciencesGS-1720GS-4182Biktarvy
→ Read original articleClinical Trials•Fierce Biotech•Apr 1, 2026

Valneva CEO on Pfizer-partnered Lyme vaccine: Why a phase 3 miss doesn’t tell the whole story
background ▾
- •VLA15 failed to meet its primary efficacy endpoint statistically, but demonstrated 73.2% efficacy at day 28 and 74.8% at day 1 post-booster, with a wide confidence interval (15.8%-93.5%) attributable to low case numbers rather than weak efficacy
- •The companies are leveraging the timing of the primary endpoint selection (day 28) as a protocol choice rather than a fixed medical threshold, plus a statistically significant p-value, to argue for regulatory flexibility with the FDA
- •FDA approval is uncertain given the current U.S. healthcare environment's increased vaccine skepticism; pediatric use restrictions are also possible pending subgroup analyses, and the regulatory outcome will depend on whether the FDA accepts the totality-of-evidence argument
ValnevaPfizerVLA15PF-07307405
→ Read original articleClinical Trials•Fierce Biotech•Apr 1, 2026

Oric chases Pfizer into phase 3 hailing prostate cancer edge, but investors send stock down
background ▾
- •Rinzimetostat's efficacy appears comparable to mevrometostat at both 400 mg and 600 mg doses, but Oric is betting competitive differentiation on superior safety profile (fewer GI, hematological, dysgeusia, and alopecia events).
- •The phase 3 dose selection of 400 mg over 600 mg reflects a safety-driven strategy rather than efficacy plateau, with the lower dose showing better tolerability and fewer dose modifications.
- •Watch for phase 3 topline data readout timing and whether Oric can clinically validate its claimed safety edge; Pfizer's mevrometostat is already in pivotal testing, establishing the timeline pressure for Oric.
Oric PharmaceuticalsPfizerBayerrinzimetostatmevrometostatNubeqa
→ Read original articleClinical Trials•Fierce Biotech•Apr 1, 2026

Drug combo treats multiple cancers with ‘paradox’ mutation in mice
background ▾
- •LSD1 and PARP1 inhibition synergistically kills BAP1-deficient cancers across four tumor types by disrupting nucleotide excision repair—a previously unrecognized BAP1 function—offering a targeted therapeutic strategy for a common but understudied oncogenic pathway
- •Both candidate drugs (seclidemstat and Lynparza) have existing human safety data, substantially reducing development risk and accelerating potential clinical trial initiation versus de novo compounds
- •Watch for industry partnership announcements and Investigational New Drug (IND) applications for the LSD1/PARP1 inhibitor combination in BAP1-mutant cancers; mesothelioma and renal cell carcinoma are likely initial indications given robust preclinical efficacy
Duke-NUS Medical SchoolNational Cancer Center SingaporeUniversity of Hawaiʻi Cancer CenterseclidemstatLynparzaolaparib
→ Read original articleClinical Trials•Fierce Biotech•Apr 1, 2026

Thousands of clinical trial sites impacted by Middle East conflict: report
background ▾
- •6.7% of active global clinical trials (4,361 studies) involving nearly 8,000 sites have been disrupted, with Phase 3 programs at highest risk due to large, long-term patient cohorts
- •All top 10 pharma companies have significant exposure in affected countries (400-500 sites each), requiring rapid contingency planning and potential deployment of advanced analytical techniques like digital twins
- •Watch for sponsor announcements on trial protocol amendments, patient safety reports, regulatory filings requesting trial modifications, and whether companies successfully implement alternative statistical approaches to maintain data integrity
Tofigh Daru Research and Engineering Company
→ Read original article