Wednesday, April 1, 2026
← Back to archivePolicy
Policy•Fierce Pharma•Mar 31, 2026

BMS, Novartis, Gilead, Iovance dinged over biologics promos in rare spate of CBER untitled letters
background ▾
- •Four major pharma companies received nearly identical CBER untitled letters for promoting unapproved efficacy endpoints (OS, PFS) in materials for approved biologics, marking the first significant cluster of CBER enforcement actions since 2018 and part of broader FDA advertising crackdown.
- •The FDA specifically objected to use of exploratory data from single-arm studies beyond approved primary endpoints and found that standard footnotes/caveats were insufficient to mitigate misleading overall messaging, establishing stricter standards for biologic promotional claims.
- •Watch for industry response and potential precedent: whether companies' 15-day responses result in compliance or escalation; whether this pattern extends to additional biologics; and whether the FDA applies similar enforcement to conventional drugs already flagged in prior untitled letters.
Bristol Myers SquibbNovartisGilead SciencesAmtagviBreyanziKymriah
→ Read original articlePolicy•KFF•Mar 31, 2026

Overview of President Trump’s Executive Actions on Global Health
background ▾
- •Trump administration initiated broad executive actions targeting global health programs and USAID operations starting day one of second term, with ongoing implementation through March 2026
- •Actions include foreign aid review and potential USAID dissolution, creating uncertainty around funding for international health initiatives and pharmaceutical access programs in developing countries
- •Pharma companies dependent on global health partnerships, international clinical trial infrastructure, and emerging market access should monitor implementation timelines and budget allocations
Policy•KFF•Mar 31, 2026
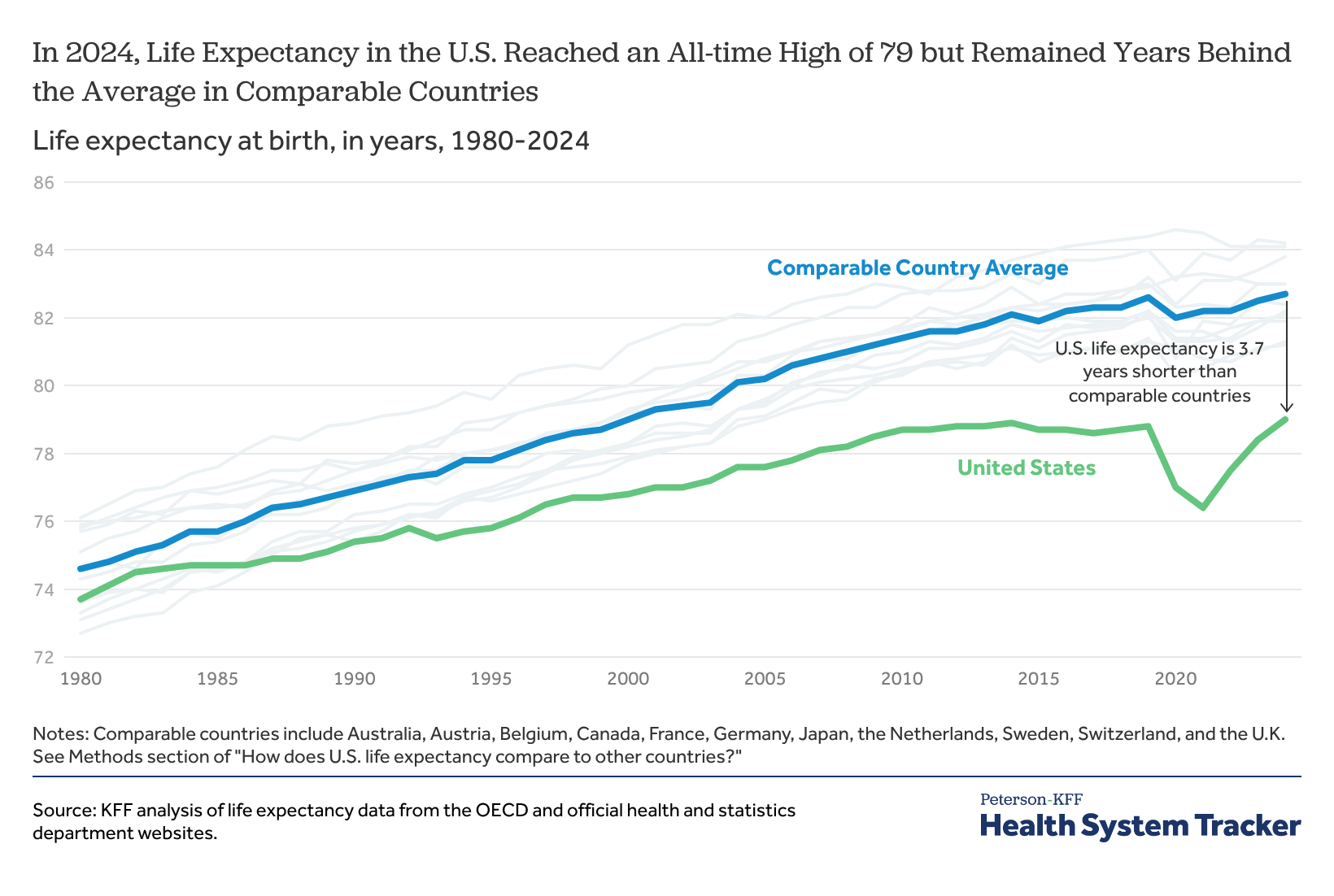
How Does U.S. Life Expectancy Compare to Other Countries?
background ▾
- •U.S. life expectancy gap versus OECD peers persists at 3.7 years despite incremental domestic improvement
Policy•Fierce Biotech•Mar 31, 2026

Fierce Biotech Layoff Tracker 2026: Lipella, IO shutter; Takeda plots 634 US job cuts
background ▾
- •Takeda is cutting 634 US jobs (247 in Massachusetts) as part of a $1.26 billion annual cost-reduction restructuring, effective July 2026 under new CEO Julie Kim
- •Multiple biotech closures and major restructurings (IO Biotech shutdown, Lipella bankruptcy, Evotec 800-person layoff) reflect sustained clinical trial failures and investor pressure on unprofitable programs
- •Monitor whether the scale of 2026 layoffs exceeds 2025 trends, and track how Takeda's restructuring under new leadership impacts its pipeline priorities and global operations
IO BiotechLipella PharmaceuticalsTakedaseralutinibINO-3107
→ Read original articlePolicy•KFF•Mar 31, 2026

Public Opinion on Prescription Drugs and Their Prices
background ▾
- •Polling data reflects public sentiment rather than market-moving developments; useful for understanding pressure on drug pricing policy but does not indicate regulatory or commercial changes
Market Access
Market Access•Fierce Pharma•Mar 31, 2026

Biogen, eyeing swift commercial tailwind, ponies up $5.6B for Apellis and its 2 approved meds
background ▾
- •Biogen acquires two revenue-generating approved drugs generating $689M in 2025 sales (Syfovre $587M; Empaveli $102M) to fill the commercial growth gap until its late-stage pipeline launches circa 2028.
- •Syfovre faces direct competition from Astellas' Izervay in a large but underpenetrated GA market of ~1.5M U.S. patients; Biogen expects mid- to high-teens annual revenue growth for both products over the next two years, signaling confidence in market expansion and competitive positioning.
- •Watch for: Biogen's Q1 2026 earnings call for updated 2026 guidance; Syfovre vs. Izervay competitive dynamics and market share trends; Empaveli's uptake trajectory in newly approved kidney indications and role as anchor for Biogen's broader nephrology franchise.
BiogenApellis PharmaceuticalsAstellas PharmaSyfovreEmpaveliIzervay
→ Read original articleMarket Access•Fierce Pharma•Apr 1, 2026

Evotec hires exec with AI experience to lead rebooted commercial team
background ▾
- •Evotec is rebuilding commercial leadership with an AI-focused executive as the company attempts to reverse falling revenues and stabilize its discovery/preclinical services business
- •Khan's appointment signals Evotec's strategic pivot toward leveraging AI and robotics capabilities to differentiate its service offerings and capture higher-value customer opportunities
EvotecIktosSchrödinger
→ Read original articleMarket Access•Fierce Biotech•Apr 1, 2026

KBP hopes to revive troubled heart drug at center of deal fraud dispute with Novo Nordisk
background ▾
- •Ocedurenone showed a 4.3 mmHg blood pressure reduction versus placebo (just below the 5 mmHg futility threshold) with p=0.002 statistical significance in the failed phase 3, creating ambiguity about whether the program was truly futile or prematurely terminated.
- •Novo alleges KBP deliberately withheld critical due diligence information about a Bulgarian trial site's data integrity issues that inflated phase 2 results, whereas KBP argues Novo's allegations are a pretext to escape a failed deal.
- •Watch for arbitration ruling in New York; potential asset recovery by KBP could enable a repositioned phase 3 in a narrower hypertension subpopulation, though funding and regulatory strategy remain unclear.
KBP BiosciencesNovo Nordiskocedurenone
→ Read original articleMarket Access•Fierce Pharma•Mar 31, 2026

Iterum initiates wind-down after failure to offload antibiotic with sluggish sales
background ▾
- •Orlynvah generated only $400,000 in sales in its launch quarter (Q3 2025) while incurring $6.5 million in operating expenses, making the commercial model unsustainable despite regulatory approval.
- •Iterum engaged with two potential acquirers but failed to complete a deal, exhausting all strategic alternatives before liquidation.
- •Watch for outcome of the April 13 high court hearing and whether Orlynvah's assets/intellectual property are acquired by another entity or abandoned entirely.
Iterum TherapeuticsOrlynvah
→ Read original articleMarket Access•Fierce Pharma•Mar 31, 2026

API supplier BASF raises prices up to 20% in response to rising energy, raw material costs
background ▾
- •BASF's 20% price increase signals material cost pressures propagating through the pharma supply chain and will likely trigger similar actions from competing suppliers and manufacturers
- •Specific critical-care and high-volume generic APIs face severe supply concentration risk: Jordan produces ~50% of global amoxicillin oral suspension and etomidate API; Jordan and Israel combined supply 73% of flumazenil API
- •Monitor: further geopolitical escalation, announcements from other major suppliers (Lonza, Siegfried, Hikal, Dr. Reddy's), and whether NHS or other payers implement emergency procurement measures or pricing negotiations
BASFLonzaSiegfriedamoxicillinetomidateflumazenil
→ Read original articleClinical Trials
Clinical Trials•Fierce Biotech•Mar 31, 2026

Obesity biotech Ambrosia scores $100M series B to take next-gen oral GLP-1 into the clinic
background ▾
- •Ambrosia secured $100M Series B funding to initiate Phase 1 trials for an oral small-molecule GLP-1 agonist with claimed best-in-class potential including low dosing, 24-hour coverage, and combinability advantages
- •The small-molecule GLP-1 space is accelerating with multiple competitors advancing: Eli Lilly's orforglipron (pending FDA approval) and Structure Therapeutics reporting 16% average weight loss, intensifying competitive pressure
- •Watch for Phase 1 clinical trial initiation and data readout; competitive positioning will depend on efficacy, dosing frequency, tolerability, and potential for combination therapy—key differentiators versus incumbent peptide therapies
Ambrosia BiosciencesBlue Owl Healthcare OpportunitiesRedmileorforglipron
→ Read original articleClinical Trials•Fierce Biotech•Mar 31, 2026

Lipella files for bankruptcy, ending dream of developing mouth inflammation drug
background ▾
- •Lipella's bankruptcy terminates the only clinical-stage program targeting symptomatic OLP, leaving a therapeutic gap in a rare oral inflammatory condition despite positive phase 2a efficacy signals
- •The delisting and cash depletion sequence illustrates how regulatory/compliance issues and funding constraints can derail biotech programs regardless of clinical promise
Lipella PharmaceuticalsLP-10 (tacrolimus oral rinse)
→ Read original articleClinical Trials•Fierce Biotech•Mar 31, 2026

BMS links up with Faro to use AI to improve clinical trial protocols
background ▾
- •BMS is systematically building enterprise-scale AI capabilities across clinical development, including protocol design automation and standardization against CSDD frameworks
- •Faro's structured digital protocol approach positions it as an enabler of transparent, reproducible AI application in trial design—a potential competitive advantage if it accelerates timelines or reduces protocol deviations
- •Success will depend on whether AI-optimized protocols demonstrably reduce trial failures, timelines, or costs; early efficacy data from this partnership should be monitored
Bristol Myers SquibbFaroEvinova
→ Read original article