Sunday, March 29, 2026
← Back to archivePolicy
Policy•KFF•Mar 27, 2026

Medicaid Enrollment and Unwinding Tracker
background ▾
- •Medicaid/CHIP enrollment peaked at 94 million during continuous enrollment; unwinding data through September 2024 shows most states completed renewals by August 2024, with significant procedural disenrollments across the program
- •This represents the largest single policy-driven shift in Medicaid eligibility in recent years, directly affecting patient access to medicines and pharmaceutical utilization rates across state programs
- •Monitor ongoing pharmaceutical utilization trends and market access strategies as data emerges on how disenrolled populations access medications through alternative coverage or cash markets
Policy•KFF•Mar 25, 2026
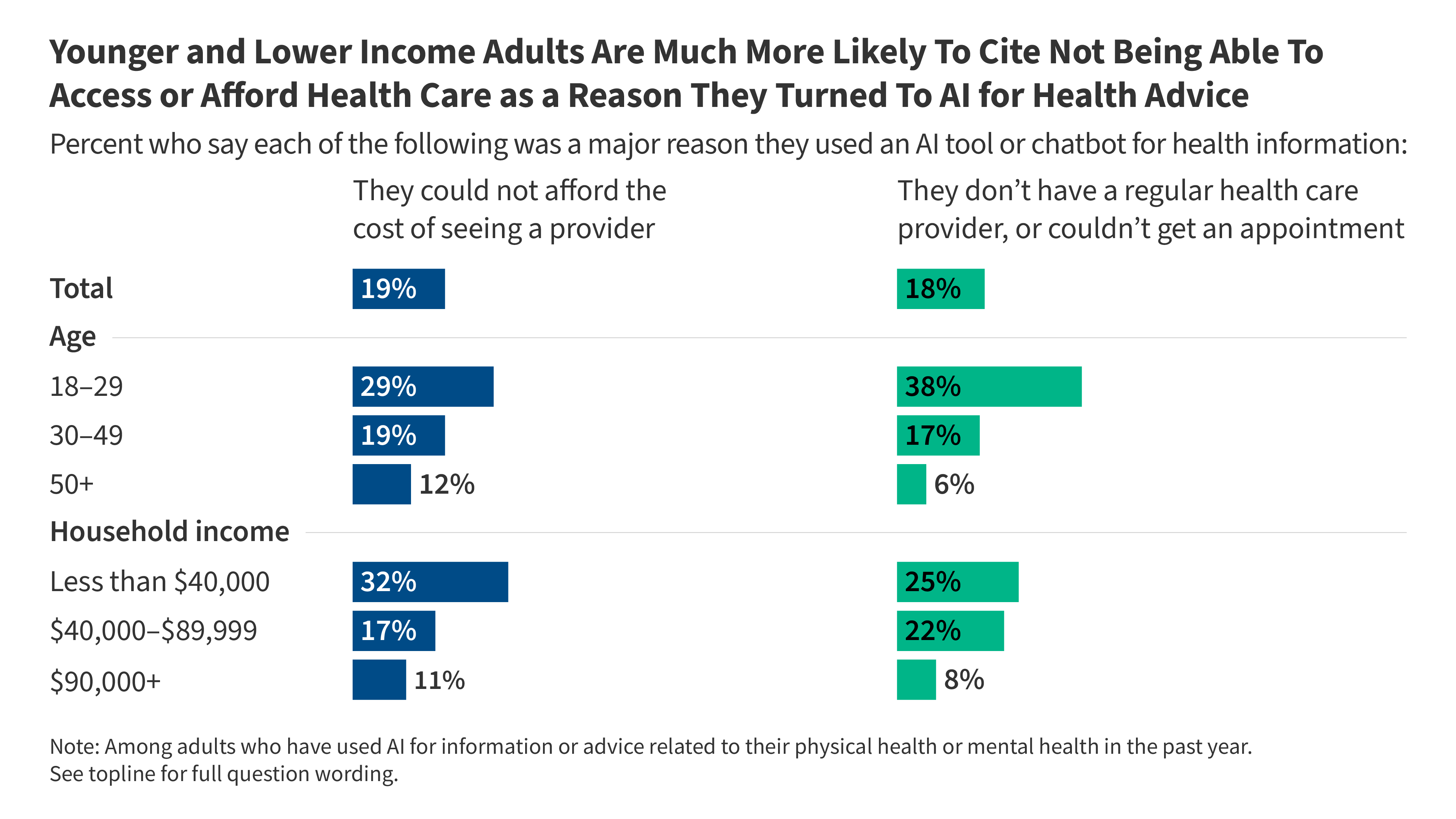
KFF Tracking Poll on Health Information and Trust: Use of AI For Health Information and Advice
background ▾
- •32% of adults now use AI chatbots for health advice, with highest adoption among ages 18-29 (36% for physical health, 28% for mental health) and uninsured/minority populations seeking mental health guidance
- •58% of adults using AI for physical health advice subsequently follow up with healthcare providers, suggesting AI serves as preliminary information source rather than replacement for clinical care
- •Watch for regulatory responses from FDA and FTC regarding accuracy standards, disclaimers, and oversight of health-specific AI chatbots as adoption accelerates and disparities become more evident
Policy•KFF•Mar 26, 2026
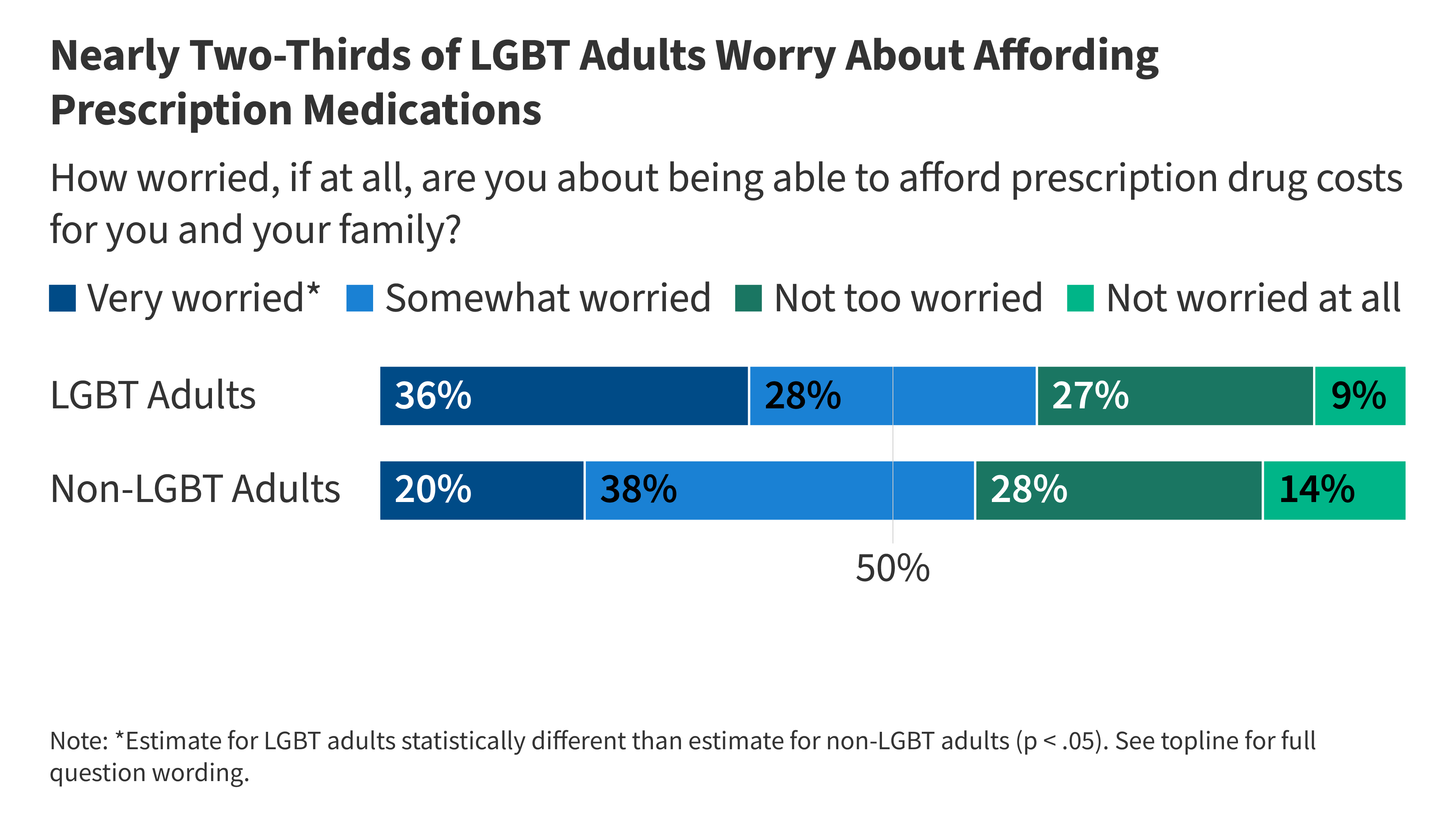
LGBT People Experience Widespread Concerns and Challenges When it Comes to Health Care Affordability
background ▾
- •LGBT adults report significantly higher rates of health care affordability challenges than non-LGBT adults, with 43% experiencing actual difficulty paying for health care and 39% struggling to afford prescription medications
- •LGBT adults are disproportionately 'very worried' about prescription drug costs (36% vs 20% of non-LGBT adults), reflecting both lower incomes and greater financial vulnerability
- •Health care affordability is positioned as a key electoral issue for the 2026 midterms, particularly given the demonstrated disparities in access and financial burden among LGBT populations
Policy•KFF•Mar 27, 2026
U.S. Global Health Country-Level Funding Tracker
background ▾
- •The U.S. appropriated $6.9 billion for bilateral global health programs in FY 2023, with the tracker covering State Department and USAID funding across 75+ countries
- •This is a public data tracking tool, not a new policy announcement, and notably excludes funding from NIH, CDC, and DoD—limiting visibility into total U.S. global health R&D investment
- •For pharma/biotech: Monitor regional funding patterns to identify emerging market opportunities and understand U.S. government priorities in vaccine, infectious disease, and maternal/child health programs
Policy•KFF•Mar 25, 2026
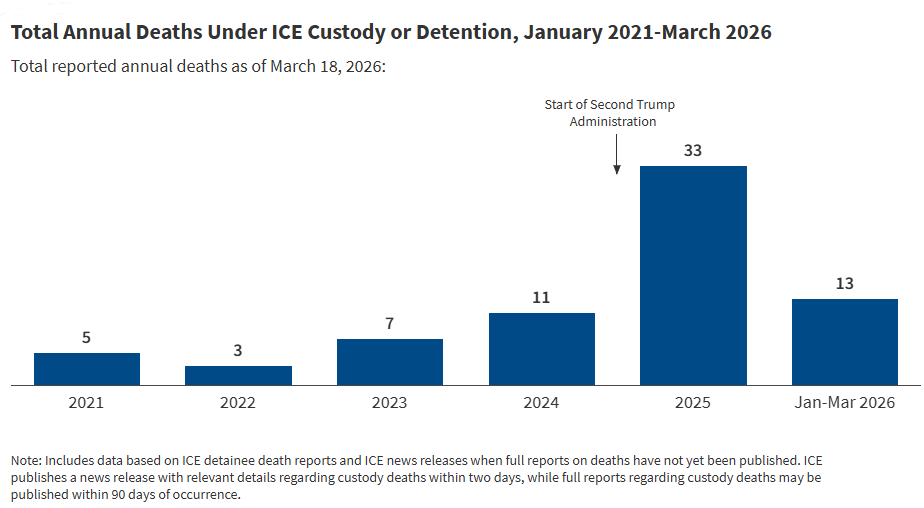
Deaths and Health Care Issues in ICE Detention Centers Under the Second Trump Administration
background ▾
- •Deaths in ICE detention increased from 11 in 2024 to 33 in 2025, with systemic health care deficiencies documented across facilities
- •Overcrowding (70% increase in detained population) raises disease transmission risks and complicates medical management of detainees with complex conditions
- •This is primarily a government health policy and detention facility oversight issue with limited direct relevance to pharmaceutical industry operations or drug pipeline decisions
Market Access
Market Access•Fierce Pharma•Mar 27, 2026

Pulse check on Lilly's GLP-1 fortunes
background ▾
- •HSBC downgrade suggests potential erosion of Lilly's previously unshakeable market position in GLP-1s and obesity despite recent stock volatility
- •Lilly maintains forward momentum with additional data on next-wave incretin candidates, indicating continued pipeline advancement
- •Broader GLP-1 market dynamics and competitive landscape are shifting, warranting close monitoring of Lilly's commercial strategy and pipeline differentiation
Eli LillyHSBCGLP-1 agonists
→ Read original articleMarket Access•Fierce Pharma•Mar 26, 2026

Egg-based drugmaker Neion Bio emerges from stealth to cook up multi-product biosimilar collab
background ▾
- •Neion Bio's novel egg-based manufacturing platform addresses a genuine bottleneck in biopharmaceutical production by offering lower capital intensity, simpler operations, and reduced development/supply costs compared to decades-old CHO cell technology
- •The company has secured its first commercial biosimilar partnership (partner unnamed) covering up to three monoclonal antibodies with revenue-sharing terms, validating market interest in alternative manufacturing approaches
- •Success will depend on whether the egg-based platform can demonstrate manufacturing reliability, scale, quality consistency, and cost advantages in real-world biosimilar production—clinical and commercial data from the partnership will be critical to watch
Neion BioAlvogenAlvotechHerceptinHumira
→ Read original articleMarket Access•Fierce Pharma•Mar 27, 2026

#FierceMadness: Elite Eight emerge in 2026 Drug Name Tournament—VOTE NOW
background ▾
- •This is a non-news entertainment feature; it has zero impact on drug development, approvals, market access, or industry strategy
- •Eight approved drugs are competing in a fan-voted naming tournament that reflects naming trends (e.g., letter 'Z' popularity, phonetic appeal)
- •No business-critical information is present—this is purely reader engagement content
Boehringer IngelheimDizal PharmaceuticalJazz PharmaceuticalsHernexeosZegfrovyModeyso
→ Read original articleMarket Access•Fierce Pharma•Mar 27, 2026

‘Health Doesn’t Need to Be Ludacris’: Bayer signs rapper-actor to multivitamin campaign
background ▾
- •This is a consumer marketing initiative with no clinical, regulatory, or significant commercial impact—routine brand advertising activity.
- •Bayer's nutritionals segment experienced a 4% sales decline in 2025, suggesting the campaign may be part of efforts to reverse underperformance in this category.
- •Monitor whether celebrity-driven wellness messaging and simplified positioning help stabilize Bayer's nutritionals sales trajectory in coming quarters.
BayerOne A Day
→ Read original articleClinical Trials
Clinical Trials•Fierce Biotech•Mar 28, 2026

Takeda clears path to FDA with phase 3 data on $4B psoriasis bet
background ▾
- •Zasocitinib demonstrated clinically meaningful superiority over placebo and apremilast in two phase 3 trials, with 61.3% and 51.9% PASI 90 response rates versus 5% and 4% for placebo and 16.8% and 15.9% for apremilast
- •Takeda claims differentiation through once-daily dosing with no food effect, positioning it favorably against Icotyde (requires fasting) but must directly compete with similarly dosed envudeucitinib and higher-efficacy IL-23 antagonists on the market
- •Regulatory approval and market entry timing for 2026, with peak sales forecast of $3-6 billion; actual market share will depend on head-to-head efficacy perception, tolerability, and payer positioning relative to entrenched competitors in the growing psoriasis market
TakedaBristol Myers SquibbJohnson & JohnsonzasocitinibapremilastSotyktu
→ Read original article