Saturday, March 28, 2026
← Back to archivePolicy
Policy•KFF•Mar 27, 2026
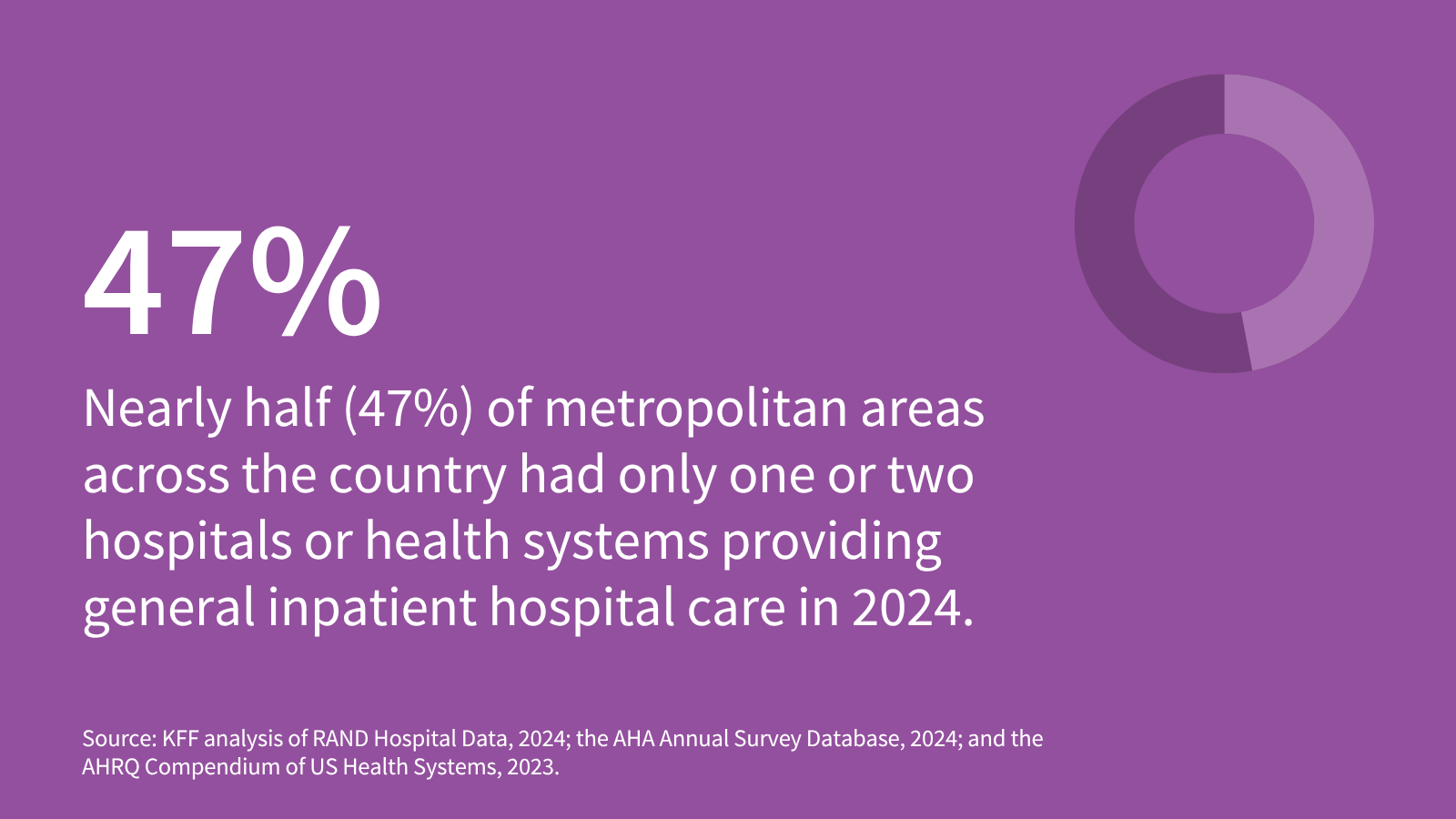
One or Two Health Systems Controlled the Entire Market for Inpatient Hospital Care in Nearly Half of Metropolitan Areas in 2024
background ▾
- •19% of metropolitan areas are controlled by a single health system and 27% by two systems; 83% of all MSAs meet the definition of highly concentrated markets under current antitrust thresholds
- •Consolidation is geographically stratified: 79% of smaller MSAs (under 200,000 population) are controlled by one or two systems, while larger metros show oligopolistic control in even the largest cities (e.g., Austin's 89% duopoly)
- •Watch for regulatory response: whether the DOJ/FTC will increase antitrust enforcement against further health system consolidation, and whether Congress will implement new antitrust thresholds or pricing regulations specific to hospital markets
HCA HealthcareAscension Healthcare
→ Read original articlePolicy•Fierce Pharma•Mar 27, 2026

Regulatory tracker: FDA signs off on Novo's once-weekly insulin Awiqli
background ▾
- •Awiqli is the first FDA-approved once-weekly basal insulin, offering significant convenience advantage over daily injections and addressing a clinically meaningful unmet need in Type 2 diabetes management
- •Phase 3 Onwards program demonstrated Awiqli's superiority in A1C reduction compared to standard daily basal insulin with acceptable safety, supporting regulatory approval on robust clinical evidence
- •Monitor Awiqli's real-world uptake and market penetration against existing basal insulin options; assess whether once-weekly formulation drives meaningful improvement in medication adherence and patient outcomes
Novo NordiskAmgenSiga TechnologiesAwiqliImdylltratecovirimat
→ Read original articlePolicy•KFF•Mar 25, 2026
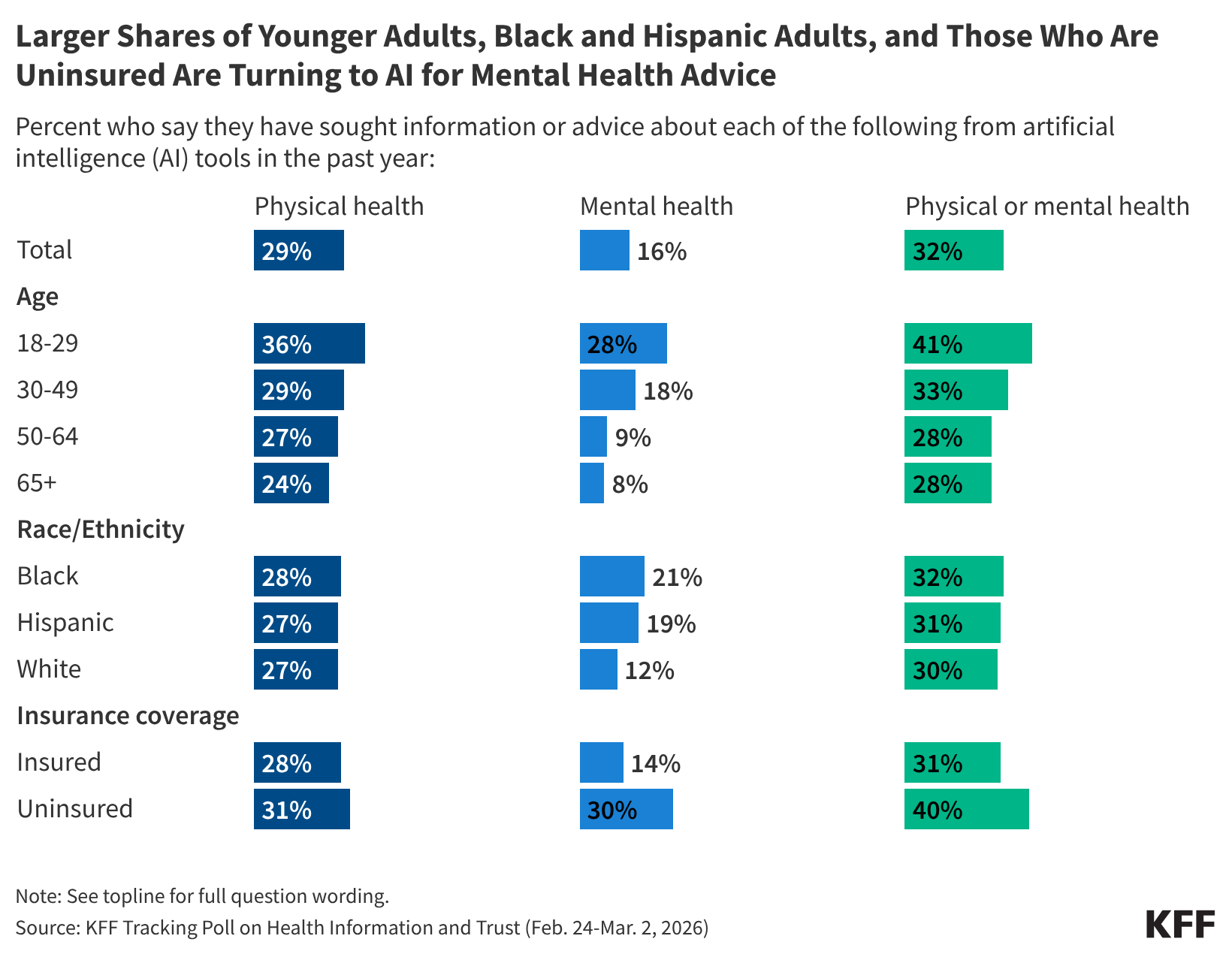
Poll: 1 in 3 Adults Are Turning to AI Chatbots for Health Information, Equaling the Share Who Use Social Media for Health
background ▾
- •One-third of American adults now use AI chatbots for health information at rates equivalent to social media use, representing a major shift in health information-seeking behavior that pharma must address in digital strategy and direct-to-consumer communications
- •Significant shares of users (42-58%) do not follow up with healthcare providers after consulting AI for health advice, creating potential patient safety risks and reducing clinical oversight that concerns regulators and pharmaceutical companies
- •Privacy vulnerabilities are substantial: 41% of AI health tool users have uploaded personal medical records to unvetted systems despite 77% public concern, signaling urgent need for FDA/FTC policy clarification on AI health tools and creating liability exposure for industry stakeholders
Policy•Fierce Biotech•Mar 27, 2026

Chutes & Ladders—Tang takes over Aurinia after FDA brouhaha
background ▾
- •Kevin Tang took over as CEO of Aurinia and installed three Tang Capital Management executives in COO, CFO, and CSO roles, replacing the previous leadership team entirely
- •Aurinia faced significant regulatory and reputational damage after a former FDA CDER director publicly questioned Lupkynis' safety and efficacy on LinkedIn, prompting an FDA investigation and lawsuit
- •Watch for Lupkynis commercialization strategy, regulatory actions from the FDA regarding the drug's labeling or market status, and whether Tang's leadership stabilizes investor confidence
Aurinia PharmaceuticalsTang Capital ManagementAbivaxLupkynisobefazimodEntyvio
→ Read original articlePolicy•Fierce Biotech•Mar 27, 2026

'We need to put some bad ideas to bed': Inside Incubate's battles for biotech on Capitol Hill
background ▾
- •Biotech industry leaders are actively lobbying against MFN price controls and IRA Medicare negotiation provisions, arguing these policies undermine innovation and competitive advantage versus China
- •Research cited by Incubate shows a 70% reduction in median VC investment in small-molecule candidates under high Medicare exposure since the IRA passed, signaling real capital flight concerns
- •Watch for FDA regulatory guidance consistency, congressional response to national security framing of biotech policy, and whether MFN codification or IRA modifications advance
IncubateForge Life Science PartnersPfizer PAVE
→ Read original articleMarket Access
Market Access•Fierce Pharma•Mar 27, 2026

Rocket plots measured trajectory for new gene therapy Kresladi after clearance to launch from FDA
background ▾
- •FDA granted accelerated approval for Kresladi, Rocket's first commercial product, based on biomarker data (neutrophil CD18/CD11a expression) with requirement for confirmatory post-marketing evidence
- •Rocket is deliberately limiting commercialization investment with a "minimal viable launch" strategy, expecting only single-digit annual patient treatments despite market potential, to fund cardiovascular gene therapy pipeline expansion
- •Watch for post-marketing registry data demonstrating clinical benefit confirmation; pricing/reimbursement strategy announcement expected closer to Q4 2027 launch; potential sale of rare pediatric disease priority review voucher
Rocket PharmaceuticalsKresladimarnetegragene autotemcel
→ Read original articleMarket Access•Fierce Pharma•Mar 27, 2026

Recordati confirms it's weighing CVC Capital buyout offer of $12.6B
background ▾
- •CVC Capital's €10.9B offer to take Recordati private values the company at a material premium and represents a major M&A event for mid-cap European pharma
- •Recordati's strong momentum—including doubled revenue over a decade and Iturisa's peak sales projection increased to €1.2B—makes the timing strategically significant for deal evaluation
- •Deal remains non-binding and contingent on due diligence, financing, and partner identification; minority shareholder approval and regulatory clearance for delisting are critical next steps
RecordatiCVC Capital PartnersEUSA PharmaIturisa
→ Read original articleMarket Access•Fierce Biotech•Mar 27, 2026

Otsuka buys Transcend in $1.2B deal, nabbing MDMA analog for psychiatric conditions
background ▾
- •TSND-201 is a non-hallucinogenic MDMA analog with FDA breakthrough designation already in phase 3 for PTSD—a rare late-stage asset in the neuroplastogen space that de-risks Otsuka's acquisition significantly
- •The $1.225B deal value ($700M upfront + $525M milestones) reflects major pharma's confidence in neuroplasticity as a paradigm shift in psychiatry, positioning Otsuka to capture first-mover advantage if phase 3 succeeds
- •Monitor phase 3 trial enrollment and data readout timelines; regulatory pathway clarity for MDMA analogs remains uncertain despite breakthrough designation, and competitive neuroplastogen programs from other sponsors could impact market positioning
Otsuka PharmaceuticalTranscend TherapeuticsTSND-201
→ Read original articleMarket Access•Fierce Biotech•Mar 26, 2026

CMR Surgical kicks off Versius Plus robot’s US commercial launch
background ▾
- •CMR Surgical has commenced U.S. commercial rollout of Versius Plus following FDA clearance, targeting the largest robotic surgery market with a cart-based platform designed for hospital and ASC integration
- •The company has achieved 45,000 patient treatments globally and holds the #2 position in robotic surgery adoption worldwide, providing validation for U.S. market entry strategy
- •Watch for: expansion of Versius Plus to additional surgical indications (colorectal, urologic, gynecologic, thoracic) and the speed of health system adoption as a meaningful alternative to Intuitive's da Vinci dominance
CMR SurgicalIntuitive SurgicalVersius Plus
→ Read original articleMarket Access•Fierce Pharma•Mar 26, 2026

As expansions come online, CDMO Hovione aims to meet industry's 'dual supply and sourcing' zeal: exec
background ▾
- •Hovione is investing $400+ million across multiple global sites with $100 million dedicated to New Jersey expansion, including new PSD-3 spray drying units and continuous tableting technology to address the fact that ~80% of new small molecules are water-insoluble
- •The company has recently onboarded 19 of the top 20 large pharma companies alongside its traditional biotech base, signaling a strategic shift toward serving larger pharmaceutical partners seeking geographically distributed supply chains
- •Watch for 2026 execution metrics: capacity utilization rates, client adoption of new spray drying and continuous manufacturing technologies, and whether regional supply chain consolidation continues post-tariff volatility
HovioneGEA
→ Read original articleClinical Trials
Clinical Trials•Fierce Biotech•Mar 27, 2026

AstraZeneca delivers dream result as phase 3 COPD hits breathe life into class rocked by flops
background ▾
- •AstraZeneca's tozorakimab achieved primary and secondary efficacy endpoints in both Oberon and Titania phase 3 trials, showing consistent benefit across former smokers, current smokers, and all eosinophil/lung function subgroups—a clean sweep that rivals failed to achieve
- •Tozorakimab's dual inhibition of ST2 and RAGE/EGFR cascades (versus single-pathway competitors) appears mechanistically differentiated and may explain superior efficacy, particularly in addressing mucus production and epithelial remodeling
- •Two additional phase 3 trials (Prospero and Miranda) are ongoing with results expected H1 2024; regulatory pathway clarity and potential label breadth remain to be determined at upcoming medical meeting presentation
AstraZenecaSanofiRegenerontozorakimabitepekimabastegolimab
→ Read original articleClinical Trials•Fierce Pharma•Mar 27, 2026

Fierce Pharma Asia—Takeda’s $1.3B reorg; India’s GLP-1 floodgates; Gilead’s $2.2B buy of a China NewCo
background ▾
- •Dizal's Zegfrovy met primary endpoint in phase 3 EGFR exon 20 NSCLC as monotherapy, outperforming chemotherapy where J&J's Rybrevant required combination therapy—positioning direct competitive challenge
- •Novo Nordisk's GLP-1/GIP/glucagon triple agonist UBT251 demonstrated superior HbA1c reduction (2.16% vs 1.77%) versus semaglutide in Chinese phase 2, with global phase 2 planned for 2025
- •Watch for: Dizal's regulatory pathway and potential label advantages; Novo's global phase 2 results on triple agonist; impact of 10+ Indian semaglutide generics on Novo's revenue and pricing strategy
TakedaNovo NordiskGilead SciencessemaglutidegamgertamigOM336
→ Read original articleClinical Trials•Fierce Biotech•Mar 27, 2026

After local tumor approval, Novocure touts positive data in metastatic pancreatic cancer
background ▾
- •PANOVA-4 phase 2 data shows 74% disease control rate with Optune Pax plus Tecentriq-based chemotherapy in metastatic pancreatic cancer versus 48% historical control—a clinically meaningful and statistically significant improvement
- •Median overall survival of 9.7 months with combination therapy versus 8.5 months in control represents a 1.2-month absolute gain, though comparison to a 2013 historical cohort introduces potential confounding variables
- •Tecentriq (atezolizumab) has no approved indication in pancreatic cancer; Novocure's next steps likely include disclosing trial design details at scientific meetings and planning path to potential metastatic pancreatic cancer indication
NovocureGenentech (Roche)Optune PaxTecentriqgemcitabine
→ Read original articleClinical Trials•Fierce Biotech•Mar 27, 2026

AstraZeneca’s in vivo CAR-T bet eradicates cancer in 3 of 5 patients, but death mars dataset
background ▾
- •ESO-T01 achieved 60% complete response rate at day 60 but all 5 patients developed grade 3+ toxicities including CRS in 4 patients, with 1 death from spinal cord compression of unclear etiology
- •The in vivo CAR-T approach shows similar adverse event rates and timing to traditional ex vivo CAR-T, potentially negating the theoretical safety advantage that justified AstraZeneca's $1B acquisition
- •Watch for durability data beyond 60 days and whether anito-cel's superior response rates (74% complete responses at 15.9 months) and established competitors make ESO-T01's clinical differentiation insurmountable
AstraZenecaEsoBiotecJohnson & JohnsonESO-T01Carvyktianito-cel
→ Read original articleClinical Trials•Fierce Biotech•Mar 27, 2026

Scientists turn pig semen extract into eye drops that kill cancer in mice
background ▾
- •Exosomes from pig semen successfully delivered anti-cancer nanoparticles across the blood-retinal barrier in mice, eliminating the need for intravitreal injections
- •The mechanism of barrier penetration in ocular tissue is distinct from reproductive tract penetration, operating through EGFR-mediated pathways
- •Retinoblastoma represents a significant unmet need in low- and middle-income countries where 80% of cases occur, but human trial timeline remains uncertain and substantial preclinical work remains
Shenyang Pharmaceutical Universitycarbon dots
→ Read original article